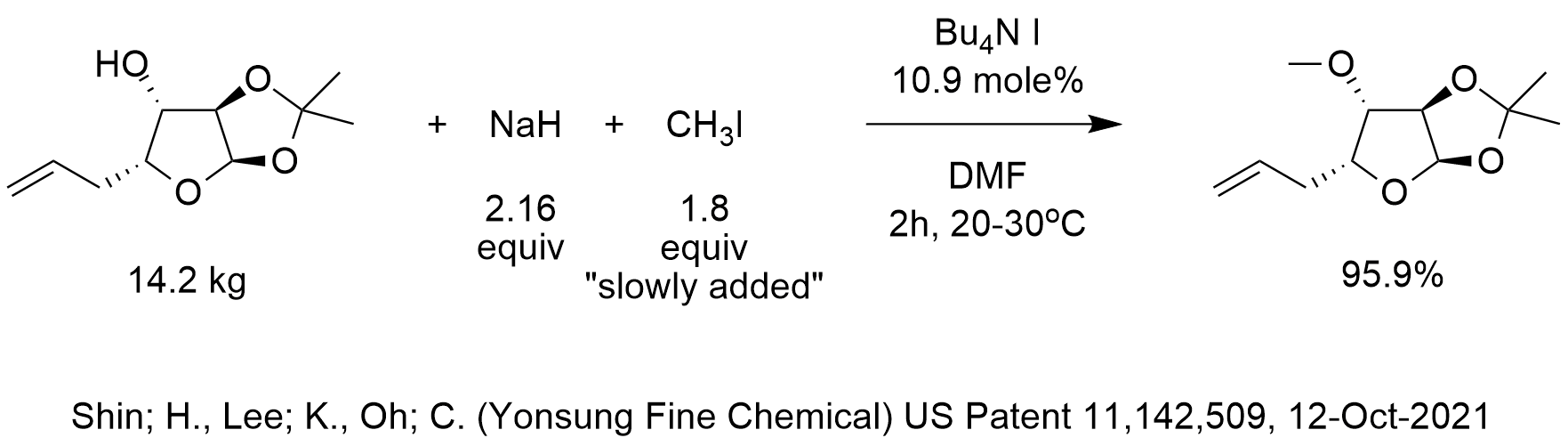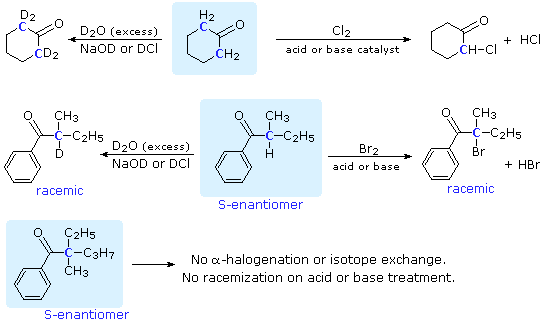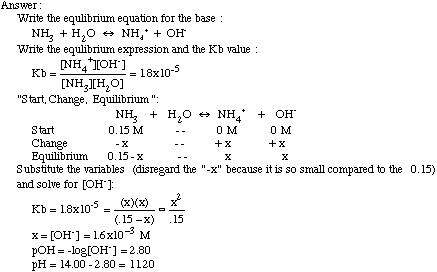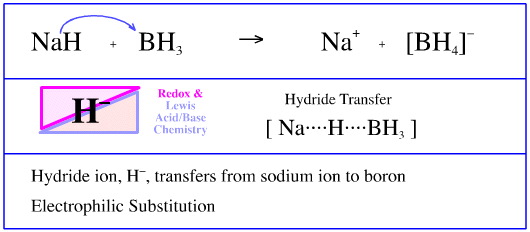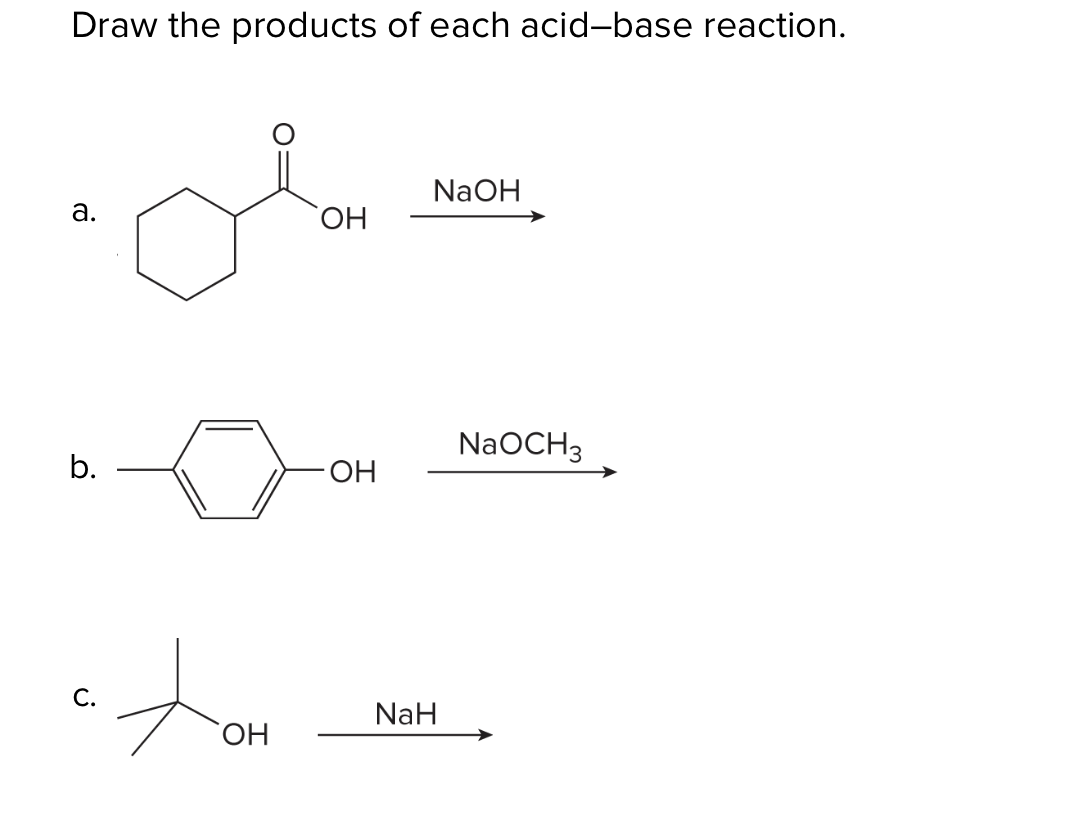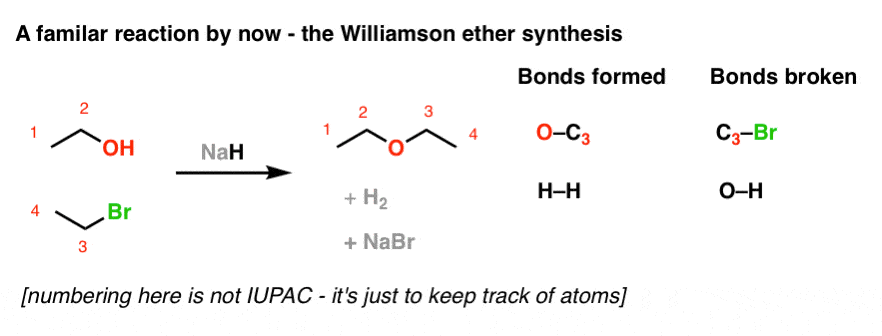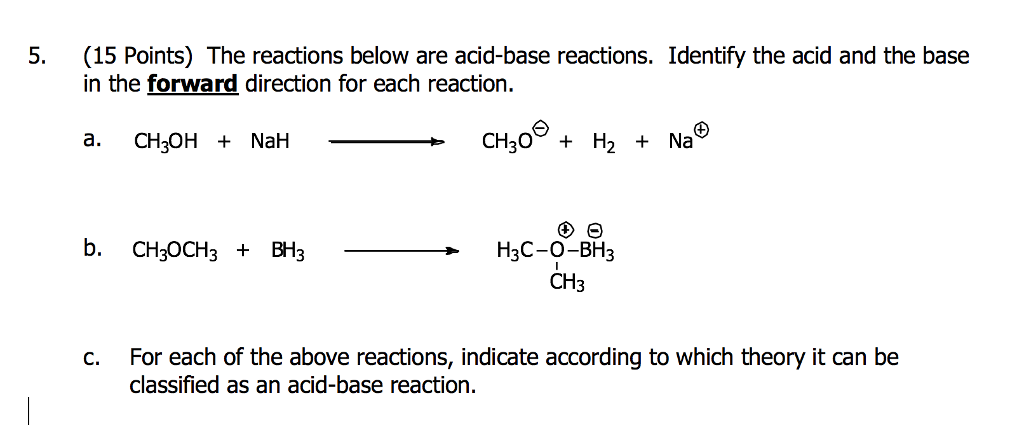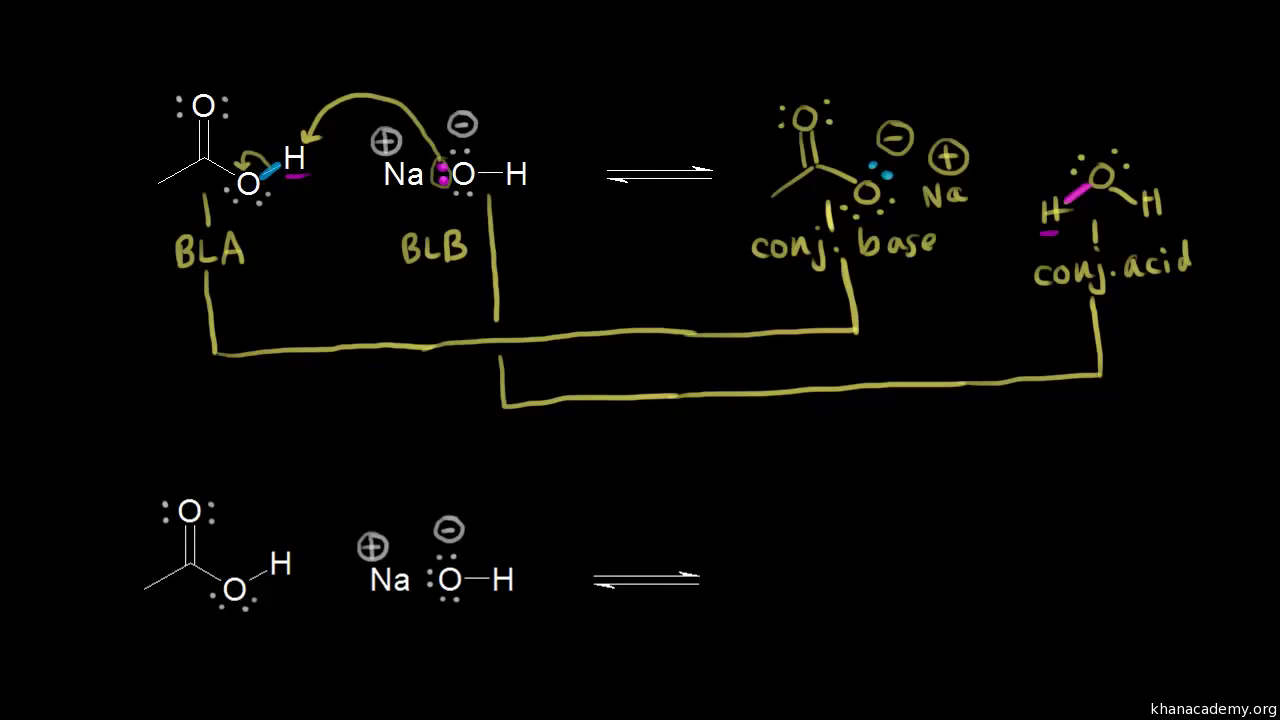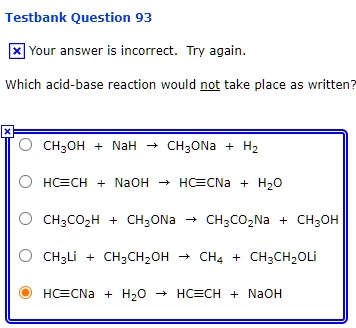
SOLVED: Testbank Question 93 Your answer incorrect Try again Which acid base reaction would not take place as written CHzOH Nah CHzONa HCCH NaOH HCCNa Hzo CH;COzH CHzONa CH;COzNa CHzOH CHzLi CHzCHzOH

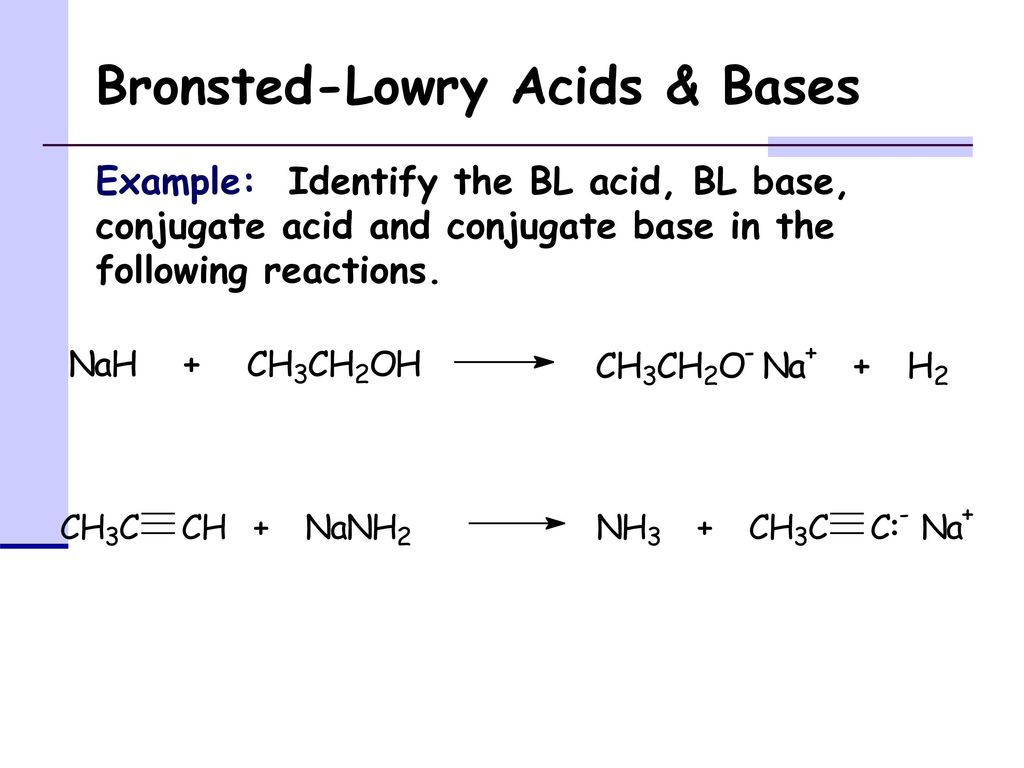
Which of the following acid-base reactions will occur? f. H-C-=CNa+CH3OH g. H-C-=CH+CH3Li h. H-C-=CH+NaH i. H-C-=CH+NaCN j. H-C-=CNa+CH3COOH

What product is formed when the following compound is treated with NaH? The following acid-base reactions were a step in a synthesis of a commercially available drug. | Homework.Study.com